In collaboration with researchers in our PET Research Centre, we are developing microfluidic approaches to the processing and synthesis of radiopharmaceuticals utilised in PET and SPECT imaging. The motivation is to enable dose-on-demand synthesis and minimise infrastructure and lab-safety requirements due to the smaller radioactive volumes processed.
We have employed particle beds and monoliths for pre-concentration. In the processing of radiopharmaceuticsl for PET imaging, fluoride generated from the cyclotron needs to be pre-concentrated prior to synthesis. We are also working with monoliths for the pre-concentration and purification of radioactive gallium from the gallium generator prior to synthesis.

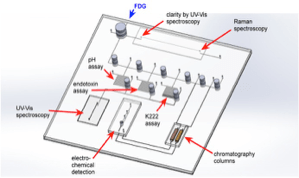
Pharmaceuticals must undergo stringent quality control (QC) before release to the patient. In the context of radiopharmaceutical for PET imaging, this includes physical, chemical, radiochemical and biological purity. Current quality control systems require an entire dose. In the context of dose-on-demand synthesis of single doses, QC tests must be minimised and automated. We are developing an integrated QC platform, featuring fibre optical absorbance spectrometers for pH, kryptant and endotoxin analysis, Raman spectroscopy for residual solvents, separation systems in conjunction with electrochemical and novel methods for radiation detection at the small scale.

Currently we are developing microfluidic devices for the production of [68Ga] radiotracers in collaboration with the East Yorkshire NHS towards GMP production, supported by the MRC.
Selected References:
- M.P. Taggart, M.D. Tarn, M.M.N. Esfahani, D.M. Schofield, N.J. Brown, S.J. Archibald, T. Deakin, N. Pamme, Lee F. Thompson, Development of radiodetection systems towards miniaturised quality control of PET and SPECT radiopharmaceuticals, Lab on a Chip, 2016, doi 10.1039/C6LC00099A
- M.D. Tarn, D. Maneuski, R. Alexander, N.J. Brown, V. O’Shea, SL. Pimlott, N. Pamme, S.J. Archibald, Positron detection in silica monoliths for miniaturised quality control of PET radiotracers, Chemical Communications, 2016, doi: 10.1039/C5CC10518H.
- P. He, B.P. Burke, G.S. Clemente, N.J. Brown, N. Pamme, S.J. Archibald, Monolith-based 68Ga processing: a new strategy for purification to facilitate direct radio labelling methods, Reaction Chemistry and Engineering, 2016, 1, 361-365. doi: 10.1039/C6RE00081A
- V. Arima, G. Pascali, O. Lade, H.R. Kretschmer, V. Hammond, P. Watts, F. De Leonardis, M.D. Tarn, N. Pamme, P. Dittrich, B. Cvetkovic, N. Vasovic, R. Duane, A. Jaksic, A. Zacheo, A. Zizzari, L. Marra, E. Perrone, P.A. Salvadori, R. Rinaldi, Radiochemistry on chip: towards dose-on-demand synthesis of PET radiopharmaceuticals, Lab on a Chip, 2013,13, 2328–2336. doi: 10.1039/c3lc00055a
- M.D. Tarn, G. Pascali, F. De Leonardis, P.A. Salvadori, P. Watts, N. Pamme, Purification of 2-deoxy-2-[18F]fluoro-D-glucose radiotracer by on-chip solid-phase extraction, Journal of Chromatrography A, 2013, 1280, 117-121. doi: 10.1016/j.chroma.2013.01.032
- F. De Leonardis, G. Pascali, P.A. Salvadori, P. Watts, N. Pamme, On-chip pre-concentration and complexation of [18F]fluoride ions via a regenerable anion exchange particles for radiochemical synthesis of Positron Emission Tomography tracers, Journal of Chromatography A, 2011, 29, 1218, 4714-4719. doi: 10.1016/j.chroma.2011.05.062